- +86 15383000851
- +86 15303238802
- admin@hebeianda.cn
Your Location:Home >Products >Biochemical Engineering >159634-47-6

Purity:99%
|
Description |
Ibutamoren (INN) (developmental code names MK-677, MK-0677, L-163,191)is a non-peptidic, potent, long acting, orally-active, and selective agonist of the ghrelin receptor and a growth hormone secretagogue, mimicking the growth hormone (GH)-stimulating action of the endogenous hormone ghrelin. It has been demonstrated to increase the release of, and produces sustained increases in plasma levels of several hormones including GH and insulin-like growth factor 1 (IGF-1), but without affecting cortisol levels. MK677 has shown to sustain activation of GH-IGF-1 Axis and increase in lean body mass but no change in total fat mass or visceral fat. It is currently under development as a potential treatment for reduced levels of these hormones, such as in children or elderly adults with growth hormone deficiency, and human studies have shown it to increase both muscle mass and bone mineral density, making it a promising therapy for the treatment of frailty in the elderly. |
|
Uses |
Growth hormone releasing factor. |
|
Safety |
Ibutamoren is a muscle building anabolic compound. It's considered safe if you don't have high blood glucose level and if you don't suffer from congestive heart failure. It's considered a boon for anti aging and it's a life longevity material. It's considered to be more beneficial than to be harmful. The optimal dose for body building is to take 25mg at bedtime for four to six weeks. It is a water retaining compound so beware of bloating. |
InChI:InChI=1/C27H36N4O5S/c1-26(2,28)25(33)29-22(18-36-17-20-9-5-4-6-10-20)24(32)30-15-13-27(14-16-30)19-31(37(3,34)35)23-12-8-7-11-21(23)27/h4-12,22H,13-19,28H2,1-3H3,(H,29,33)/t22-/m1/s1
The preparation of the Merck Growth Horm...
There are disclosed certain novel compou...
![N-[(R)-[(1,2-Dihydro-1-methanesulfonylspiro[3H-indole-3,4'-piperidin]-1'-yl)carbonyl]-2-(phenylmethyloxy)ethyl]-2-[(1,1-dimethyl-ethoxy)carbonyl]amino-2-methyl-propanamide](/upload/2023/8/bb5dd76b-2587-40df-a7da-3766aeaf1b8e.png)
N-[(R)-[(1,2-Dihydro-1-methanesulfonylspiro[3H-indole-3,4'-piperidin]-1'-yl)carbonyl]-2-(phenylmethyloxy)ethyl]-2-[(1,1-dimethyl-ethoxy)carbonyl]amino-2-methyl-propanamide

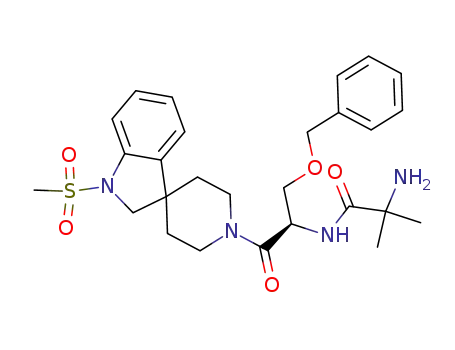
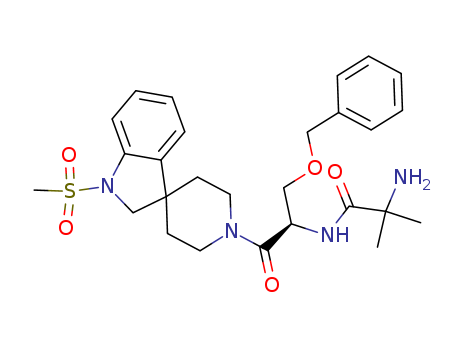
Ibutamoren
| Conditions | Yield |
|---|---|
|
With
methanesulfonic acid;
In
ethanol;
at 35 - 40 ℃;
Yield given;
|
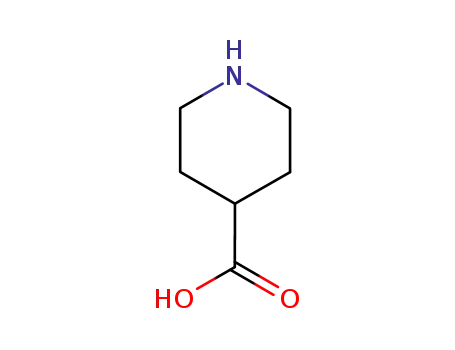
isonipecotic acid


Ibutamoren
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 11 steps
1: 97 percent / K2CO3 / H2O / 58 h / 22 °C
2: (COCl)2 / toluene; dimethylformamide / 16 h / 18 °C
3: 94 percent / H2, DIEA, thioanisole / Pd/C / toluene / 22 h / 20 °C / 2068.6 Torr
4: 99 percent / TFA / CH2Cl2 / 17 h / 35 °C
5: NaBH4 / toluene / 0.5 h / -2 °C
6: DIEA / tetrahydrofuran / 2 h / 5 - 8 °C
7: 93 percent / H2 / Pd-C / ethanol / 5 h / 65 °C / 2068.6 Torr
8: DCC, HOBt / H2O; various solvent(s) / 5 h / Ambient temperature
9: MsOH / ethanol / 7.5 h / 35 - 40 °C
10: DCC, HOBt / various solvent(s) / 2 h / Ambient temperature
11: MsOH / ethanol / 35 - 40 °C
With
sodium tetrahydroborate; oxalyl dichloride; methanesulfonic acid; methyl-phenyl-thioether; hydrogen; potassium carbonate; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine; dicyclohexyl-carbodiimide; trifluoroacetic acid;
palladium on activated charcoal;
In
tetrahydrofuran; ethanol; dichloromethane; water; N,N-dimethyl-formamide; toluene;
|
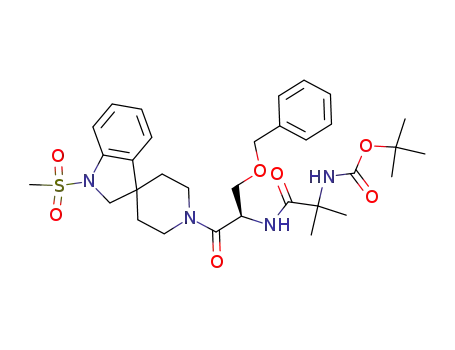
N-[(R)-[(1,2-Dihydro-1-methanesulfonylspiro[3H-indole-3,4'-piperidin]-1'-yl)carbonyl]-2-(phenylmethyloxy)ethyl]-2-[(1,1-dimethyl-ethoxy)carbonyl]amino-2-methyl-propanamide

isonipecotic acid
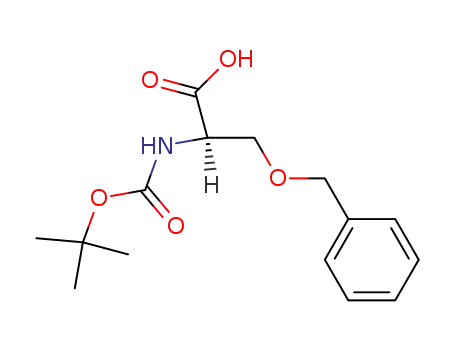
N-Boc-D-serine(Bzl)-OH
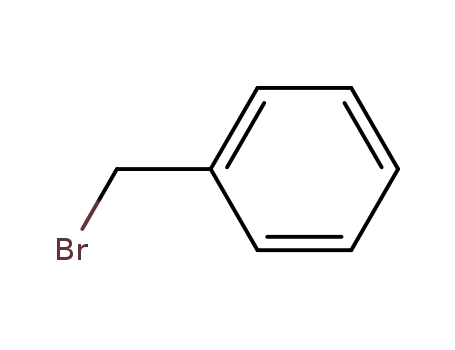
benzyl bromide
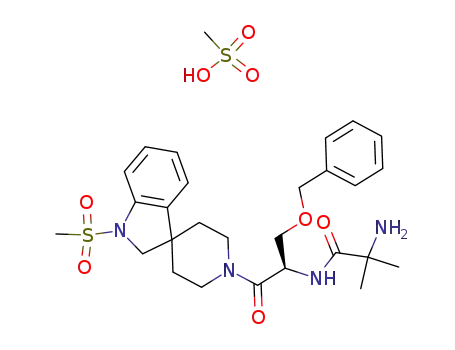
MK-677
CAS:868844-74-0
CAS:52-90-4
CAS:79517-01-4
CAS:52232-67-4