- +86 15383000851
- +86 15303238802
- admin@hebeianda.cn

pd_meltingpoint:204-206 °C (dec.)
Appearance:yellow crystalline powder
Purity:99%
|
Chemical Properties |
yellow crystalline powder |
|
Uses |
antiarrhythmic, alpha2 agonist, anticonvulsant, antiinflammatory, antibacterial, antifungal, antitrypanosomal, antineoplastic, immunostimulant |
|
General Description |
A highly potent and selective oxysterol EBI2 (GPR183) agonist (Kd) = 450 pM in a saturation binding assay, and EC50 = 140 pM over EC50 = 2.1 nM for its enantiomer, 7β,25-OHC, in a GTP-γS binding assay). Dose-dependently suppresses forskolin-induced cAMP accumulation in an EBI2-expressing SK-N-MC/CRE-β-galactosidase cell line (IC50 = 2 nM), but not in control cells. Stimulates migration of LPS-activated spleen B-cells and anti-CD3/CD28-activated CD4+ T-cells in a dose-dependent manner. In addition, pharmacological inhibition of its biosynthesis in vivo by Clotrimazole, a CYP7B1inhibitor, promotes the migration of adoptively transferred pre-activated B cells to the T/B boundary, mimicking the phenotype of pre-activated B cells in EBI2-deficient mice. |
|
Biochem/physiol Actions |
An alkaloid with weak antibiotic properties. Substrate for MDR efflux pumps. Antimicrobial activities of berberine is potentiated by the MDR inhibitor 5′-methoxyhydnocarpin (5′-MHC). Berberine upregulates the expression of Pgp in hepatoma cells. Treatment with berberine potentially results in the reduced accumulation of chemotherapeutic drugs. |
|
Safety Profile |
Poison by intraperitoneal route.Slightly toxic by ingestion. Mutation data reported. Whenheated to decomposition it emits toxic vapors of NOx andCl-. |
|
Purification Methods |
Berberine chloride crystallises from water to give the dihydrate. The anhydrous salt may be obtained by recrystallisation from EtOH/Et2O, wash the crystals with Et2O and dry them in a vacuum. The iodide has m 250o(dec) (from EtOH). [Perkin J Chem Soc 113 503 1918, Kametani et al. J Chem Soc(C) 2036 1969, Beilstein 27 I 515, 27 II 567.] |
InChI:InChI=1/C20H18NO4.ClH/c1-22-17-4-3-12-7-16-14-9-19-18(24-11-25-19)8-13(14)5-6-21(16)10-15(12)20(17)23-2;/h3-4,7-10H,5-6,11H2,1-2H3;1H/q+1;
The fluorescence enhancement of berberin...
A modularly convergent and divergent str...
The total synthesis of berberine and sel...
Berberine hydrochloride is a natural alk...
The present invention belongs to the fie...
![2-(6-(2-(2-(1,3-dioxolan-2-yl)-3,4-dimethoxyphenyl)acetyl)benzo[d][1,3]dioxol-5yl)ethyl pivalate](/upload/2023/8/e7833ee5-19ec-435b-8f80-41f61f9ba6f8.png)
2-(6-(2-(2-(1,3-dioxolan-2-yl)-3,4-dimethoxyphenyl)acetyl)benzo[d][1,3]dioxol-5yl)ethyl pivalate

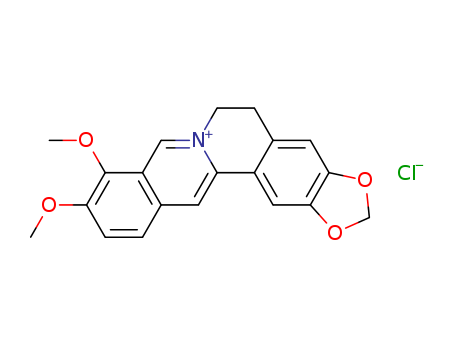
berberine chloride
| Conditions | Yield |
|---|---|
|
With
ammonium chloride;
In
ethanol; water;
at 110 ℃;
for 24h;
Concentration;
Solvent;
|
89.4% |
|
With
ammonium chloride;
In
ethanol; water;
at 90 - 110 ℃;
for 56h;
|
82% |
C20H17NO4


berberine chloride
| Conditions | Yield |
|---|---|
|
With
ammonium chloride;
In
water;
at 150 ℃;
for 40h;
Inert atmosphere;
Sealed tube;
|
37% |
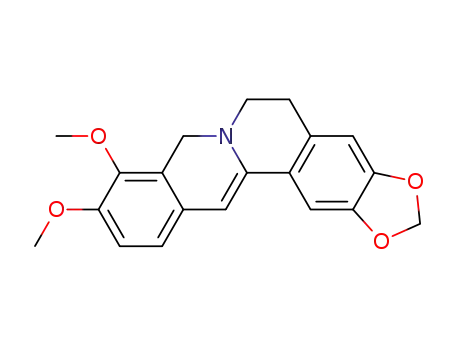
7,8-dihydroberberine
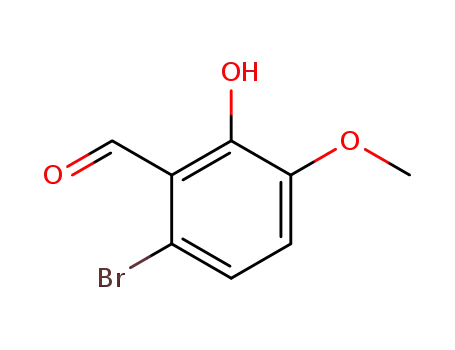
6-bromo-2-hydroxy-3-methoxybenzaldehyde
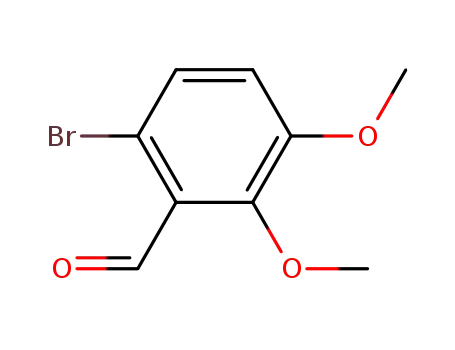
6-bromo-2,3-dimethoxybenzaldehyde
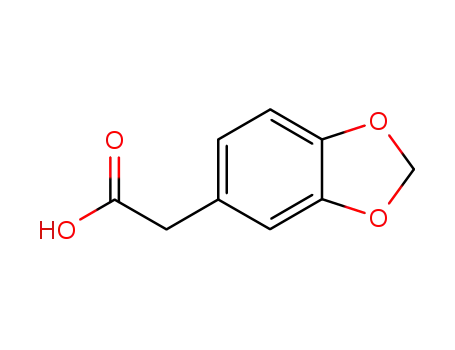
1,3-benzodioxole-5-acetic acid
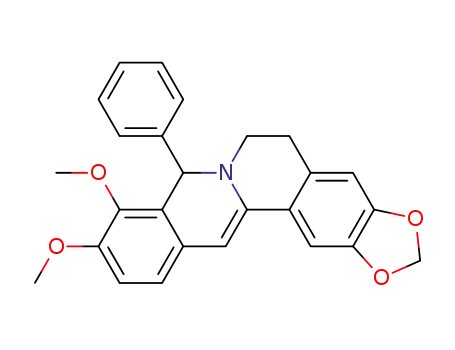
9,10-dimethoxy-8-phenyl-5,8-dihydro-6H-[1,3]dioxolo[4,5-g]isoquino[3,2-a]isoquinoline

7,8-dihydroberberine
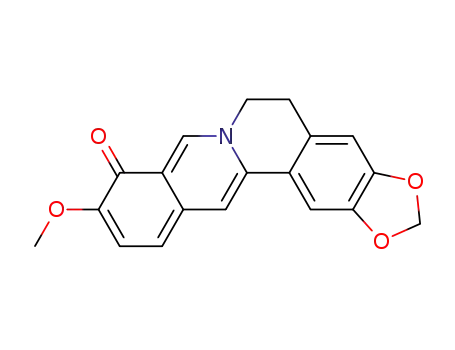
berberrubine
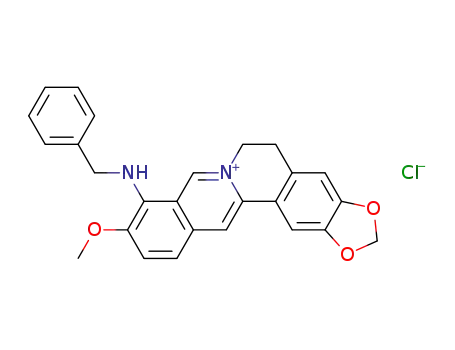
N-benzylberberine chloride
CAS:23239-88-5
CAS:73-78-9
CAS:53-39-4
CAS:137-58-6